About CFTR-France database
CFTR-France: a patient-related database integrating sequence variations, genotypes and phenotypes stored in diagnostic laboratories specialized in the analysis of CFTR variation
CFTR-France has been developed since 2012 with the aim to collect, store and process any category of variants identified in the CFTR gene, thanks to the collaboration of 10 French laboratories with high expertise in the molecular analyses of this gene and the financial support of the parents association Vaincre la Mucoviscidose.
Since 2013, CFTR-France has been collaborating with the French Clinical Registry of patients affected with cystic fibrosis, via the association Vaincre la Mucoviscidose.
CFTR-France is a 'LSDB' (Locus Specific DataBase) dedicated to sequence variations of the CFTR gene associated with diverse clinical phenotypes, the most severe of which is Cystic Fibrosis (CF, OMIM #219700). CF is the most common life-shortening inherited autosomal recessive disorder in populations of European descent.
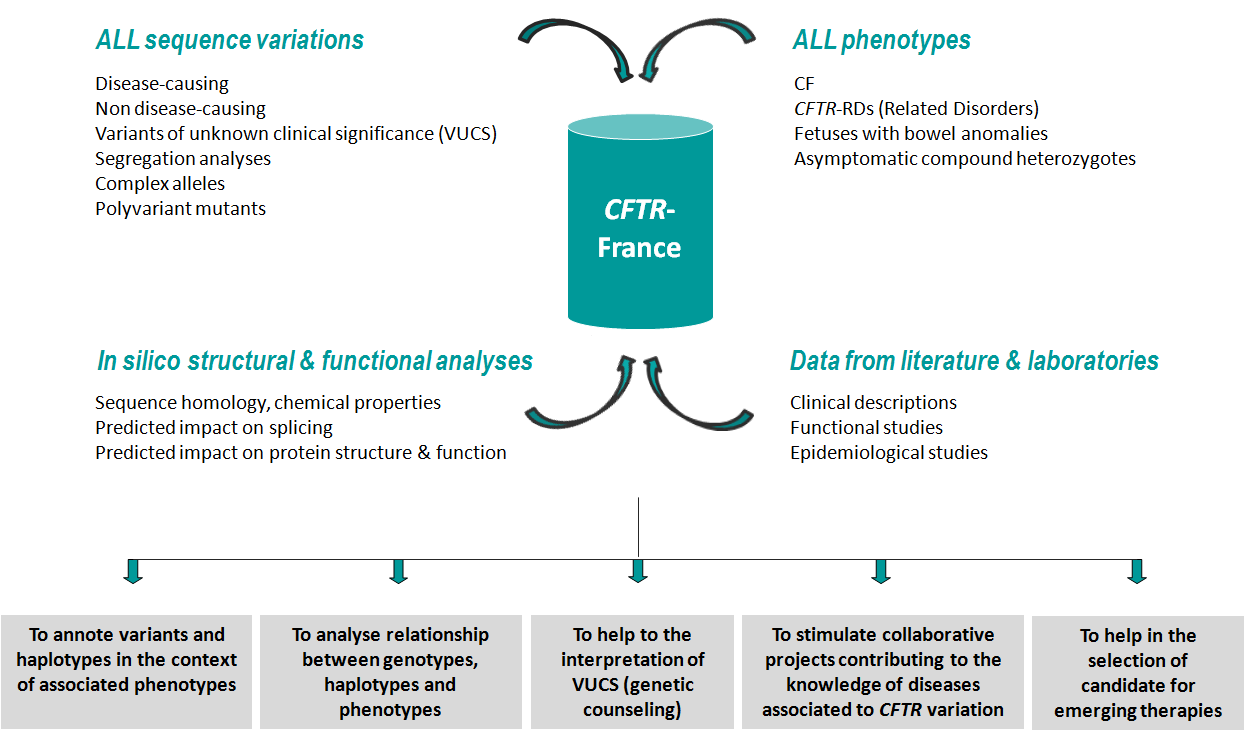
The specificity of CFTR-France is to compile any category of variations (disease-causing, non disease-causing and variants of unknown clinical significance)
that have been identified by nine specialized laboratories in the following situations:
The database includes the main clinical data of these individuals, genetic information from familial segregation studies, and, various variant annotations (frequency in patient and control populations, sequence homology, predicted or experimentally assessed functional impact, etc), allowing the analysis of genotype/phenotype relationships.
CFTR-France is meant to be a source of information on rare CFTR variants, regularly updated, and to contribute to a better knowledge on their clinical significance. The project consists in:
CFTR-France is not intended to allow direct submission of variants.
- Patients affected with cystic fibrosis (CF)
- Patients presenting milder phenotypes named CFTR-related disorders (CFTR-RD)
- Fetuses with abnormal ultrasonography (echogenic bowel, )
- Newborns with pending or inconclusive diagnosis
- Asymptomatic individuals carrying at least one sequence variation on each CFTR gene (i.e. carrying two variations in trans).
The database includes the main clinical data of these individuals, genetic information from familial segregation studies, and, various variant annotations (frequency in patient and control populations, sequence homology, predicted or experimentally assessed functional impact, etc), allowing the analysis of genotype/phenotype relationships.
CFTR-France is meant to be a source of information on rare CFTR variants, regularly updated, and to contribute to a better knowledge on their clinical significance. The project consists in:
- Collecting (retrospectively) genetic data from nine French laboratories that are recognized as experts with the CFTR genetic diagnosis,
- Performing regular review of epidemiological data, experimental data, literature, and central databases on CFTR variations,
- Performing research on the clinical and functional impact of CFTR variants.
CFTR-France is not intended to allow direct submission of variants.
Coordinating Team
- Directors of the project: Pr. Mireille CLAUSTRES and Dr. Caroline RAYNAL
- Software developers: Dr. Corinne BAREIL and Dr. Souphatta SASORITH
- Data curators: Dr. Souphatta SASORITH, Dr. Corinne BAREIL, Dr. Anne BERGOUGNOUX and Dr. Caroline RAYNAL
The coordinating team in Montpellier assumes the following tasks:
- To develop electronic formats for data submission by collaborators,
- To collect, reformat, treat and curate data sent by the laboratories,
- To create appropriate bioinformatics tools for storage, analysis and retrieval,
- To implement the database infrastructure,
- To update the database contents (approximately four times/year),
- To retrieve, on request, all data on a variant identified in problematic diagnosis situations e.g. fetal suspicion of CF, genetic counseling for couples [data restricted to the French network of diagnostic laboratories],
- To organize each year a workshop bringing together the laboratories participating to CFTR-France database, representatives of the parents association 'Vaincre la Mucoviscidose', and representatives of the French Clinical CF Registry. The achievements of the year are presented by the coordinating team,
- To write the annual meeting report, which is available to all partners.
Board of Collaborators
- The Board of Collaborators includes the laboratories who accepted to contribute and share genetic or clinical data and to bring their experience and expertise.
Its task is to contribute and take decisions about future developments and orientations of the database, research projects and collaborative works to solve difficulties in the interpretation of CFTR genetic variations.
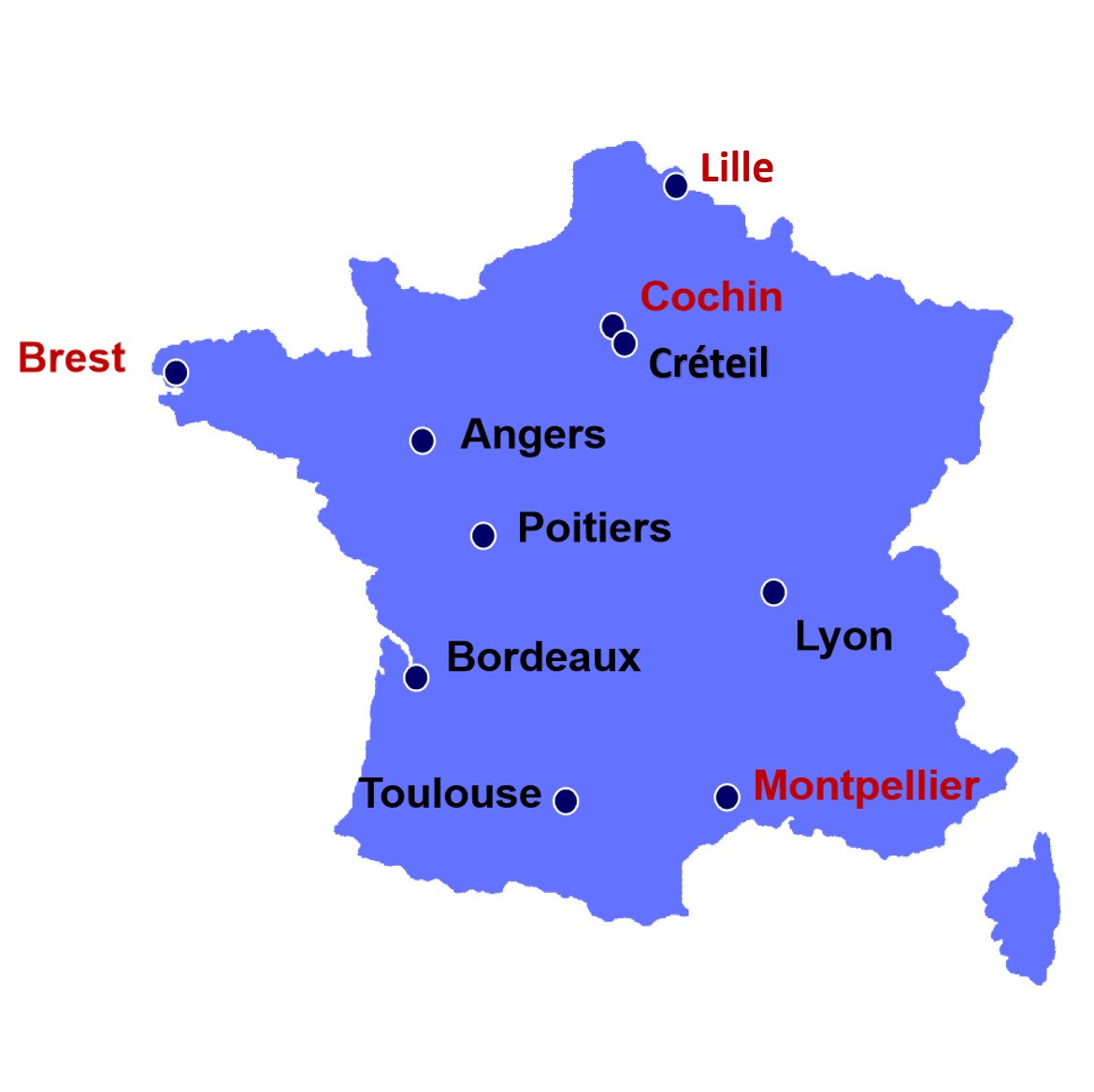
| PARTICIPANTS | REGIONS |
| A. Diagnostic laboratories with recognized expertise in CFTR analysis | National Reference Laboratories |
|---|---|
| Dr. Emmanuelle GIRODON, Pr. Thierry BIENVENU | Paris-Broca-Cochin-Hôtel Dieu |
| Dr. Marie-Pierre AUDREZET, Pr. Claude FEREC | Brest |
| Dr. Caroline RAYNAL, Pr. Mireille CLAUSTRES | Montpellier | National Expert Laboratories |
| Dr. Adrien PAGIN, Dr. Guy LALAU | Lille |
| Dr. Alix DE BECDELIEVRE, Dr. Pascale FANEN | Créteil |
| Dr. Marie-Claire MALINGE | Angers |
| Dr. Marie-Pierre REBOUL, Dr. Patricia FERGELOT | Bordeaux |
| Dr. Fabienne DUFERNEZ, Pr. Alain KITZIS | Poitiers |
| Véronique GASTON, Dr. Eric BIETH | Toulouse |
| Dr. Céline RENOUX, Dr. David CHEILLAN | Lyon | B. Parents Association 'Vaincre La Mucoviscidose' |
| Antoine BESSOU (French Registry for CF patients) | Paris |
Database curation
- Anonymized raw data are sent as excel files by diagnostic laboratories to Montpellier, the unique curation center. Disparate data are reshaped to be recorded into a common database after a thorough verification
by the curators of nomenclature, genotypes, allele assignation, other variations in cis or in trans, associated clinical data, extent of variant detection and techniques, and other collected information
(e.g. geographical origins).
Only accurate information is stored in CFTR-France, which implies multiple exchanges between the curators and the participating laboratories.
The CFTR-France was created in 2008 at Montpellier University by Mireille CLAUSTRES, Corinne BAREIL, Marie Des GEORGES, Corinne THEZE and Caroline RAYNAL [IURC, Institut Universitaire de Recherche Clinique, Inserm U827].
It is now maintained by Corinne BAREIL, Souphatta SASORITH, Anne BERGOUGNOUX, Caroline RAYNAL and Mireille CLAUSTRES [Laboratoire de Génétique Moléculaire de maladies rares, CHU de Montpellier - Site Unique de Biologie R+3, PhyMedExp INSERM U1046, CNRS UMR 9214, Univ Montpellier].
The curation of the database is partially funded by "Vaincre la Mucoviscidose".
The conception of the software and the bioinformatics management of the database are not funded by external grants.
It is now maintained by Corinne BAREIL, Souphatta SASORITH, Anne BERGOUGNOUX, Caroline RAYNAL and Mireille CLAUSTRES [Laboratoire de Génétique Moléculaire de maladies rares, CHU de Montpellier - Site Unique de Biologie R+3, PhyMedExp INSERM U1046, CNRS UMR 9214, Univ Montpellier].
The curation of the database is partially funded by "Vaincre la Mucoviscidose".
The conception of the software and the bioinformatics management of the database are not funded by external grants.
CFTR-France constitutes the intellectual property of the director and curators of the database. Any unauthorized copying, storage or distribution of this material without written permission from the curators would lead to copyright and database right infringement with possible ensuing litigation. Copyright © 2017. All rights reserved. For further details, please refer to Directive 96/9/EC of the European Parliament and of the Council of March 11 (1996) on the legal protection of databases.
The public information provided by CFTR-France is for scientific purposes only, not for clinical usage.
Whilst CFTR-France exercises all reasonable care to ensure that the database and data contained therein are of high quality, it makes no warranty, expressed or implied, as to the accuracy or completeness thereof or that this database or the data contained therein, is fit for a particular purpose such as the diagnosis, genetic counseling and treatment of patients. The Directors, Curators and Collaborators cannot be held responsible for any consequences arising out of any inaccuracies, omissions or misusages.
Citing CFTR-France:
Claustres M, Thèze C, des Georges M, Baux D, Girodon E, Bienvenu T, Audrezet MP, Dugueperoux I, Férec C, Lalau G, Pagin A, Kitzis A, Thoreau V, Gaston V, Bieth E, Malinge MC, Reboul MP, Fergelot P, Lemonnier L, Mekki C, Fanen P, Bergougnoux A, Sasorith S, Raynal C, Bareil C. CFTR-France, a national relational patient database for sharing genetic and phenotypic data associated with rare CFTR variants. Hum Mutat. 2017 Oct;38(10):1297-1315. doi: 10.1002/humu.23276. PMID: 28603918References related to CFTR-France:
Bergougnoux A, Taulan-Cadars M, Claustres M, Raynal C. Current and future molecular approaches in the diagnosis of cystic fibrosis. Expert Rev Respir Med. 2018 May;12(5):415-426. doi: 10.1080/17476348.2018.1457438. PMID: 29580110Atelier sur le Diagnostic Moléculaire de la Mucoviscidose (13-15 September 2018, Baillargues)
Postal address
Laboratoire de Génétique Moléculaire de maladies rares, CHU de Montpellier - Site Unique de Biologie R+3PhyMedExp INSERM U1046, CNRS UMR 9214, Univ Montpellier
371 Avenue du Doyen Gaston Giraud
34295 Montpellier cedex 5
FRANCE
